| CAS Number | 64-04-0 |
|---|---|
| Molecular Formula | C8H11N |
| Molecular Weight | 121.183 |
| InChI Key | BHHGXPLMPWCGHP-UHFFFAOYSA-N |
| LogP | 1.41 |
| Synonyms |
|
Applications:
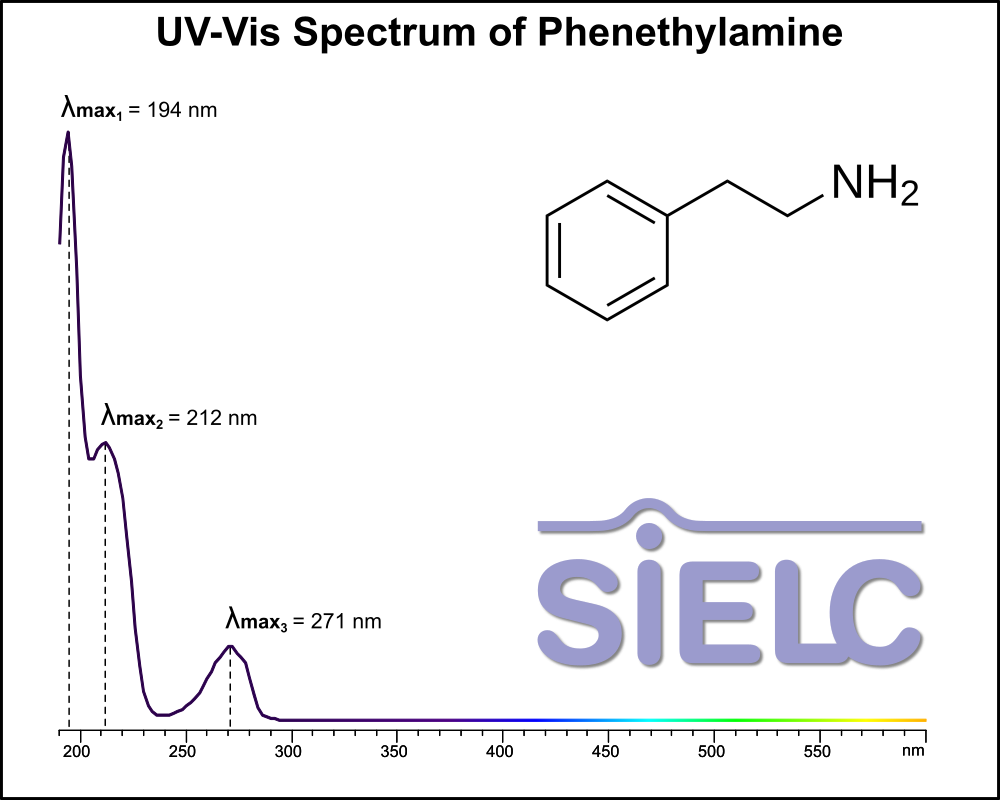
Uv-Vis Spectrum of Phenethylamine
February 25, 2026
Access the UV-Vis Spectrum SIELC Library
If you are looking for optimized HPLC method to analyze Phenethylamine check our HPLC Applications library
For optimal results in HPLC analysis, it is recommended to measure absorbance at a wavelength that matches the absorption maximum of the compound(s) being analyzed. The UV spectrum shown can assist in selecting an appropriate wavelength for your analysis. Please note that certain mobile phases and buffers may block wavelengths below 230 nm, rendering absorbance measurement at these wavelengths ineffective. If detection below 230 nm is required, it is recommended to use acetonitrile and water as low UV-transparent mobile phases, with phosphoric acid and its salts, sulfuric acid, and TFA as buffers.
For some compounds, the UV-Vis Spectrum is affected by the pH of the mobile phase. The spectra presented here are measured with an acidic mobile phase that has a pH of 3 or lower.

HPLC Separation of Histamine and Phenethylamine on Primesep C Column
August 7, 2010

Histamine is a basic compound that acts like neurotransmitter. Phenethylamine is a natural amine alkaloid psychoactive compound with stimulant effects. It forms by enzymatic decarboxylation of phenylalanine, and also acts as neurotransmitter. Both compounds are separated in one HPLC run on Primesep C and Primesep 200 columns by reversed-phase cation-exchange mechanism. Primesep C provides shorter retention time and histamine and phenethylamine can be separated within 5 minutes. Primesep C and Primesep 200 columns do not require ion-pairing reagent as it is attached to the surface of Primesep silica gel. Method can be used for analysis of these two compounds in plasma, blood and urine with UV, ELSD, CAD or LC/MS detection.
Application Column
Primesep C
Column Diameter: 3.2 mm
Column Length: 50 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Phenethylamine

HPLC Separation of Histamine and Phenethylamine on Primesep 200 Column
June 12, 2004

Histamine is a basic compound that acts like neurotransmitter. Phenethylamine is a natural amine alkaloid psychoactive compound with stimulant effects. It forms by enzymatic decarboxylation of phenylalanine, and also acts as neurotransmitter. Both compounds are separated in one HPLC run on Primesep C and Primesep 200 columns by reversed-phase cation-exchange mechanism. Primesep C provides shorter retention time and histamine and phenethylamine can be separated within 5 minutes. Primesep C and Primesep 200 columns do not require ion-pairing reagent as it is attached to the surface of Primesep silica gel. Method can be used for analysis of these two compounds in plasma, blood and urine with UV, ELSD, CAD or LC/MS detection.
Application Column
Primesep 200
Column Diameter: 3.2 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
Phenethylamine