| CAS Number | 10549-76-5 |
|---|---|
| Molecular Formula | C16H36N |
| Molecular Weight | 242.470 |
| InChI Key | DZLFLBLQUQXARW-UHFFFAOYSA-N |
| LogP | 1.04 |
| Synonyms |
|
Applications:
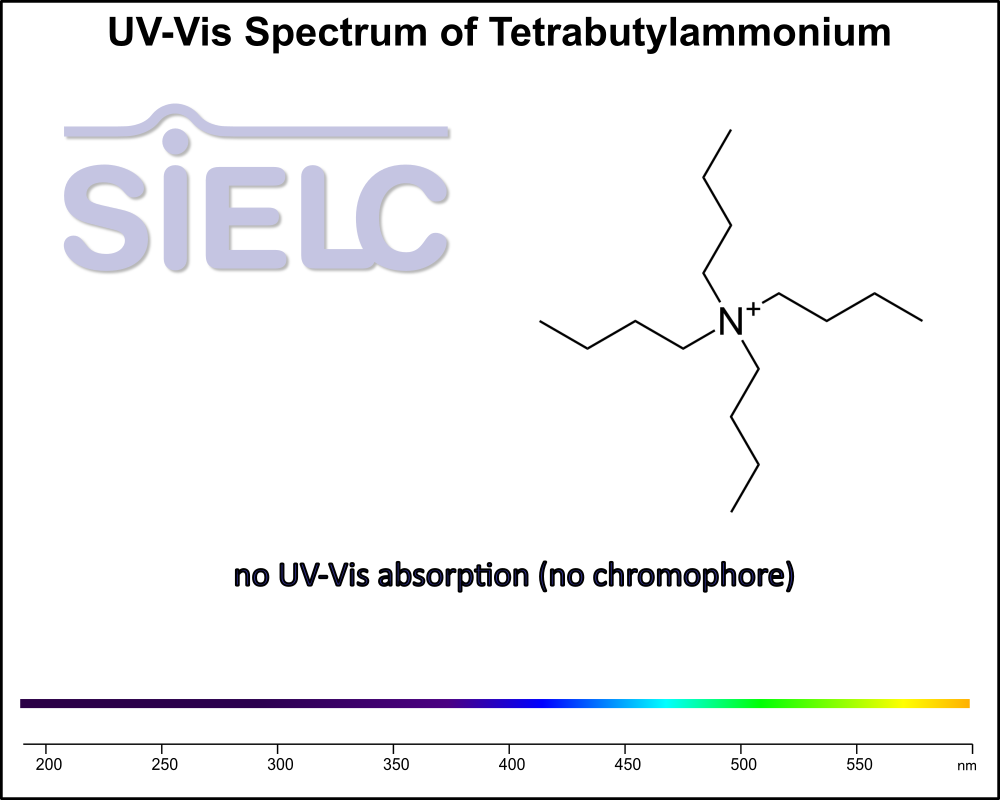
Uv-Vis Spectrum of Tetrabutylammonium
March 31, 2026
Access the UV-Vis Spectrum SIELC Library
If you are looking for optimized HPLC method to analyze Tetrabutylammonium check our HPLC Applications library
For optimal results in HPLC analysis, it is recommended to measure absorbance at a wavelength that matches the absorption maximum of the compound(s) being analyzed. The UV spectrum shown can assist in selecting an appropriate wavelength for your analysis. Please note that certain mobile phases and buffers may block wavelengths below 230 nm, rendering absorbance measurement at these wavelengths ineffective. If detection below 230 nm is required, it is recommended to use acetonitrile and water as low UV-transparent mobile phases, with phosphoric acid and its salts, sulfuric acid, and TFA as buffers.
For some compounds, the UV-Vis Spectrum is affected by the pH of the mobile phase. This analysis was done with the mobile phase made of 20% acetonitrile and 80% water.

Separation of Sodium, Tetramethylammonium and Tetrabutylammonium on Primesep 200 Column
July 7, 2011
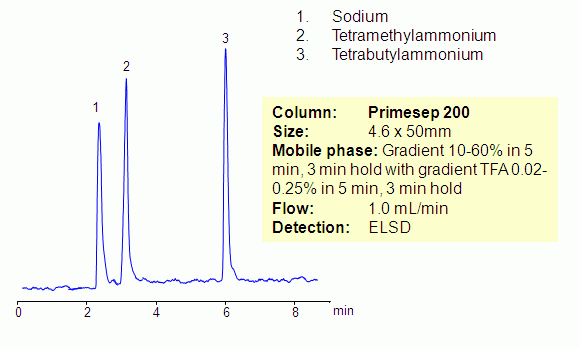
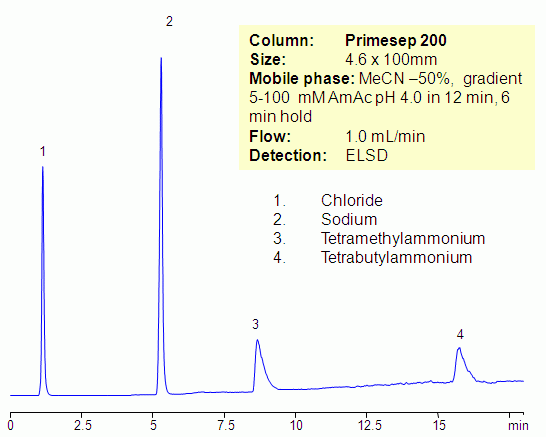
Hydrophilic and hydrophobic quaternary amines, along with sodium ion, were separated by mixed-mode chromatography on a Primesep 200 column. Mechanism of retention for sodium and tetramethylammonium ions is cation exchange, while the tetrabutylammonium ion is retained by combination of reversed-phase and cation-exchange mechanisms. All three compounds are not UV-active and monitoring is done by ELSD/CAD.
| Column | Primesep 200, 4.6×100 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmAc pH4.0 |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Ions, Hydrophilic, Ionizable, Quaternary amines |
| Analyzing Compounds | Sodium, Chloride, Tetramethylammonium, Tetrabutylammonium |
Application Column
Primesep 200
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsTetrabutylammonium
Tetramethylammonium

Separation of Quaternary Amines by Reverse-Phase Mechanism
January 16, 2004
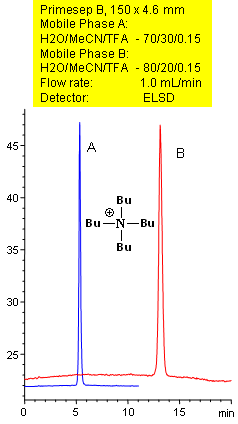
Primesep B separates quaternary amines, such as t-butylamine with symmetrical peak shape by a combination of reversed-phase and ion-exclusion mechanisms. The embedded basic functional group on the stationary phase shields the underlying silanols to prevent peak tailing. Excellent peak shape results with a mass spec compatible mobile phase of water, acetonitrile (MeCN, ACN) and trifluoracetic acid (TFA) with evaporative light scattering detection (ELSD).
| Column | Primesep B, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | TFA |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Ions, Hydrophilic, Ionizable, Quaternary amines |
| Analyzing Compounds | t-butylamine |
Application Column
Primesep B
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options