|
When selecting a reverse phase column, an analyst can choose among C30, C18, C8, C4, or C1. The longer the alkyl chain, the stronger the observed hydrophobic interaction. By analogy, we provide several columns with the same hydrophobicity, but different strength of ion-exchange to cover a broad range of interaction with various basic compounds. To achieve this, several types of acid residues are embedded in the columns stationary phase.
|
Cation Exchange
Column Type |
pKa Value of
Embedded
Carboxylic Acid |
Separates Basic Compounds |
|
Primsep 500
Primesep C
Primesep 200
Primesep 100
Primesep A
|
pKa = 5.0
pKa = 3.5
pKa = 2.0
pKa = 1.0
pKa = 0
|
Strong bases, metals
Strong bases, dibases
Strong bases, dibases
Weak, medium bases, AA, metals
Weak bases, AA, metals
|
 |
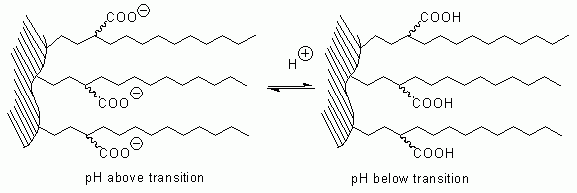
The embedded acidic functional group can be in an ionized form, or in a non-ionized form, depending on the pH of the mobile phase. In order to get retention by ion-exchange mechanism on Primesep® columns, the pH of the mobile phase should be selected close to, or above, the transition value of embedded acidic groups. Below the transition pH value, the column behaves as a regular reverse-phase column with an embedded non-ionized polar group. By changing pH of the mobile phase one can switch ion-exchange mechanism on and off.
We call this concept SWITCH Phase® Technology.
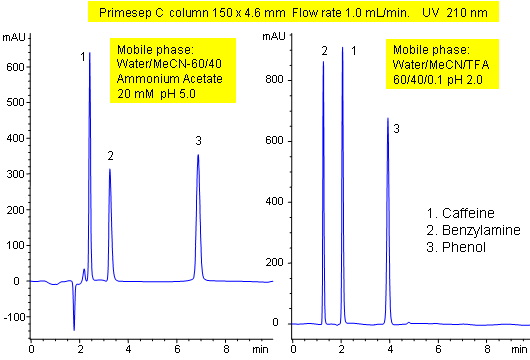
Primesep C is the easiest column to switch from the ionized form, where ion-exchange interaction contributes to the retention of basic compounds, to non-ionized form with the hydrophobic interaction as main retention mechanism. The transition pH for this column is around 4.0. This value depends not only on the pH of the mobile phase, but also on the concentration of the organic modifier. To suppress the ion-exchange in the mobile phase, the pH should be selected between 1.0 and 2.5 by using strong acids such as phosphoric, sulfuric, TFA, perchloric acid. To enhance ion-exchange property of the column, the pH should be selected between 3.5 to 7.0 by using ammonium acetate, ammonium formate or potassium phosphate as a buffer.
|
|


