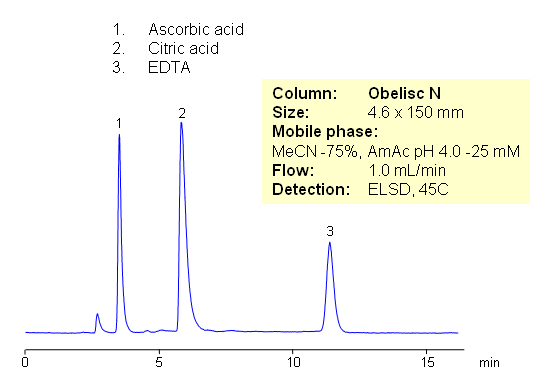
Citric acid, ascorbic acid, and EDTA are commonly used in food and pharmaceutical industry as preservatives. These compounds are very polar in nature. They are weak organic acids with limited UV activity. Retention and separation is achieved on HILIC mixed-mode Obelisc N column. All three compounds are retained by combination of strong HILIC and strong anion-exchange mechanisms. Separation can be monitored by ELSD, LC/MS, UV or Corona CAD. In contrast to other HILIC column, Obelisc N has two ionizable groups basic and acidic which provide ion-exchange interaction in addition to hydrophilic interaction. This allows to use less acetonitrile for HILIC separation.
| Column | Obelisc N , 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmAc pH 4.0 |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Acid, Vitamin B₆, Hydrophobic, Ionizable |
| Analyzing Compounds | Citric acid, ascorbic acid and EDTA |
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsEDTA (Ethylenediaminetetraacetic Acid)
Vitamin C





