HPLC Method for Analysis of Melamine on Primesep 200 by SIELC Technologies
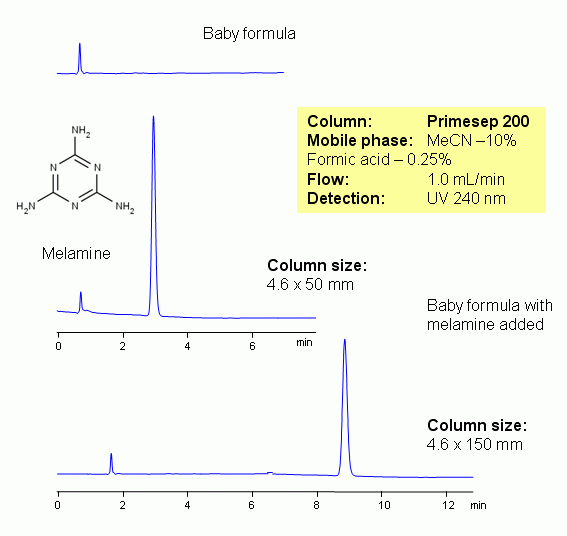
Melamine (1,3,5-triazine-2,4,6-triamine) is an organic base with pKa of 5. It is only slightly soluble in water. Melamine is a trimer of cyanamide. Melamine is a metabolite of pesticide cyromazine. Cyromazine is converted to melamine in plants. Melamine is used in production of plastics. In 2007 it was determined that melamine caused hundreds of pet deaths due to pet food contamination. Prior to these reports, melamine had been regarded as non-toxic or minimally toxic. However, because of the presence of the melamine in wheat gluten added to mass-produced dog and cat foods, it is the most likely cause. There is some data proving that melamine and cyanuric acid form a complex that show much higher toxicity.
It was found recently that melamine was added to various products (including milk formulas) to artificially inflate the reading for protein levels. Formula milk has not, until now, been tested for melamine, because regulators did not suspect this ingredient might be added. Indeed, it is this high nitrogen level – 66% nitrogen by mass – in melamine that gives it the analytical characteristics of protein molecules. A simple and fast HPLC method for determination of melamine is developed. Melamine is well retained on short (50 mm) Primesep columns. The retention is attributed to reverse phase and cation-exchange mechanism. Method uses acetonitrile-water based mobile phase with formic acid as an acidic modifier and Primesep 200 column. Method can be used in determination of melamine in food, milk, baby formulas, pet food, and other products by HPLC by direct injection or with preliminary protein clean up by ACN precipitation and filtration. Both MS (mass spectroscopy) detection and UV (ultra violet) detection at 240 nm can be effectively used to quantitate the presence and amounts of melamine with this method.
High Performance Liquid Chromatography (HPLC) Method for Analysis of Melamine
| Column | Primesep 200, 4.6 x 150 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN – 10% |
| Buffer | Formic Acid – 0.25% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 240nm |
| Class of Compounds | Hydrophobic, Ionizable |
| Analyzing Compounds | Melamine |
Application Column
Primesep 200
Column Diameter: 4.6 mm
Column Length: 150 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended
UV Detection





