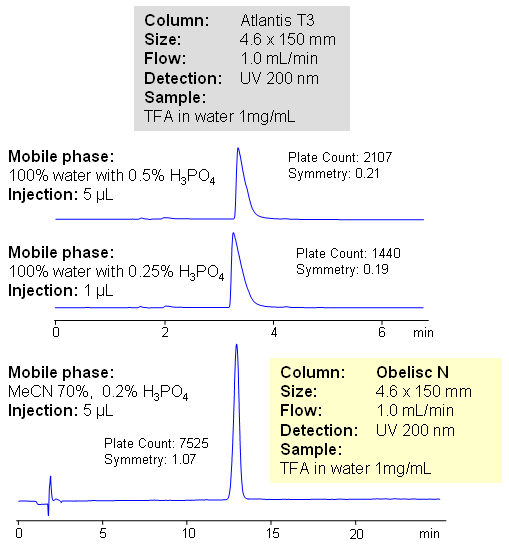
Trifluoroacetic acid (TFA) is a strong carboxylic acid, with pKa of 0.3. TFA is widely used in chemistry as reagent and as counter-ion for basic drugs and other compounds. TFA is very polar in nature, and has low UV activity. It is often required to quantitate trifluoracetic acid in various formulations. HPLC method for determination of trifluoroacetic acid on Obelisc N column provides a viable alternative for quantitation of TFA. TFA is retained by ion-exchange mechanism on mixed-mode HILIC column. Relative comparison shows high loadability of the method. Obelisc N columns show much better peak shape and retention than traditional HPLC columns (Atlantis T3 from Waters). Perfect peak symmetry is achieved by adjusting amount of acetonitrile, buffer pH and buffer concentration. TFA is monitored by low UV (200 nm). Alternatively trifluoroacetic acid can be analyzed by ESLD with ammonium formate in the mobile phase. TFA forms non-volatile salt with ammonia, thus giving ability to quantitate it.
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select options




