| CAS Number | 22839-47-0 |
|---|---|
| Molecular Formula | C14H18N2O5 |
| Molecular Weight | 294.307 |
| InChI Key | IAOZJIPTCAWIRG-QWRGUYRKSA-N |
| LogP | -2.7 |
| Synonyms |
|
Applications:
HPLC Separation of Amino Acids on Obelisc R Column
October 4, 2007
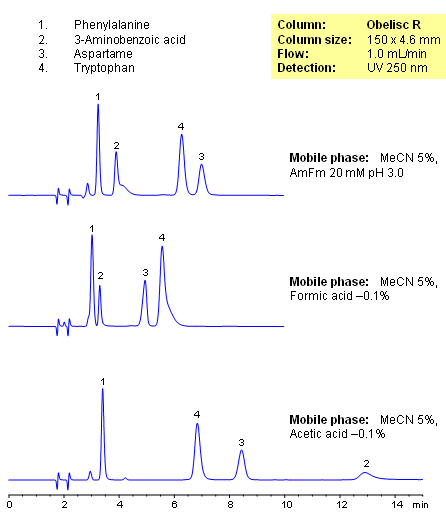
Closely related compounds like amino acids can be separated on an Obelisc R column by various buffers depending on the amount of baseline separation required. By choosing different buffers, the separation between compounds can be adjusted based on application needs, especially those that require low organic concentration in the mobile phase. UV detection at 250nm.
| Column | Obelisc R, 4.6×250 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 5/95% |
| Buffer | AmFm |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 250 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements, Amino acid |
| Analyzing Compounds | Amino acids |
Application Column
Obelisc R
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsAspartame
Phenylalanine
Tryptophan

Bufferless Ion Separation (BLIS™) Chromatography of Aspartame: Organic Modifier Effect
January 30, 2005
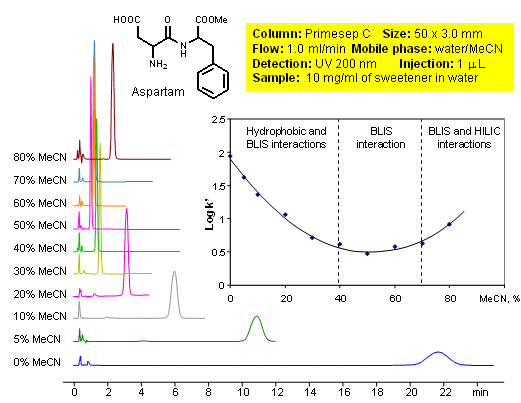
An organic modifier effect of changing acetonitrile percentage the mobile phase is demonstrated on a Primesep C HPLC column in a bufferless ion (BLIS™) separation. With decreasing percent acetonitrile, the artificial sweetener, Aspartame, first deceases in retention time and then increases. The changing mobile phase composition changes the retention mechanism from hydrophobic and BLIS™interactions, to BLIS™ alone , to BLIS™ and HILIC (hydrophilic liquid chromatography) interactions. Aspartame is composed of the two amino acids, aspartic acid and phenylalanine as the ethyl ester. Mobile phases composed of water and acetonitrile (MeCN, ACN,) and ultraviolet (UV) detection was used.
Application Column
Primesep C
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsNutrasweet