Applications:
Retention Characteristics of Betaine in Mixed-Mode HPLC
May 5, 2005
The retention of the zwitterion betaine on Primesep 200 demonstrates the tunability of Primesep columns. Retention of betaine is altered by acid type, amount of acid in the mobile phase, and the amount of organic solvent. Typical reversed-phase columns do not show this tunability with simple mass spec compatible mobile phases of water, acetonitrile (MeCN, ACN) and trifluoracetic acid (TFA) with evaporative light scattering detection (ELSD).
Application Column
Primesep 200
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsQuaternary Amines
Zwitterion

Cetylpyridinium Methods with Good Efficiency and Peak Symmetry
February 11, 2004
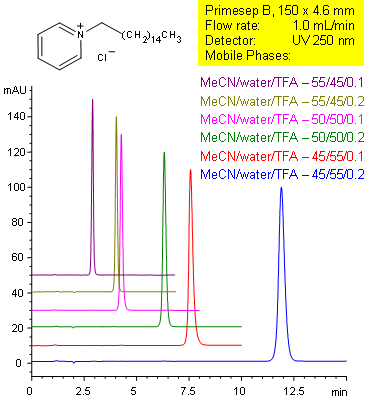
Primesep B separates tertiary amines, such as cetylpyridinium with symmetrical peak shape by a combination of reversed-phase and ion-exclusion mechanisms. The embedded basic functional group on the stationary phase shields the underlying silanols to prevent peak tailing. Retention time can be changed by changing either organic content or acid content in the mobile phase. C18 reversed-phase columns do not typically show this tuning ability with acid content. Excellent peak shape results with a mass spec compatible mobile phase of water, acetonitrile (MeCN, ACN) and trifluoracetic acid (TFA) with UV detection at 250 nm.
| Column | Primesep B, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | TFA |
| Flow Rate | 1.0 ml/min |
| Detection | UV 210nm |
| Class of Compounds |
Surfactant, Hydrophobic, Ionizable |
| Analyzing Compounds | Cetylpyridinium Chloride |
Application Column
Primesep B
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsCetylpyridinium Chloride
Pyridinium Ion
Quaternary Amines

Separation of Quaternary Amines
September 11, 2003
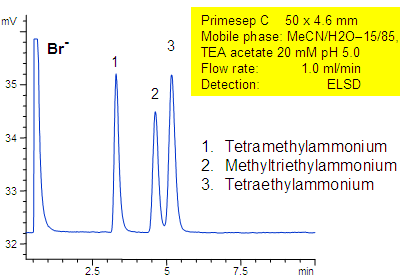
Primesep C separates a mixture of quaternary amines by a combination of cation exchange, complex formation, and hydrophic interactions. Methyltriethylammonium, tetraethylammonium, and tetramethylammonium cations as well as bromide counter ion are separated on a short 50 mm column. The separation uses a mobile phase mixture of water, acetonitrile (MeCN, ACN) and triethylamine acetate with evaporative light scattering detection (ELSD).
| Column | Primesep C, 4.6×50 mm |
| Mobile Phase | MeCN/H2O |
| Buffer | TEA acetate |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Quaternary Amines, Ionizable, Hormone |
| Analyzing Compounds | Methyltriethylammonium, tetraethylammonium, Tetramethylammonium |
Application Column
Primesep C
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsMethyltriethylammonium Bromide
Quaternary Amines
Tetraethylammonium
Tetramethylammonium