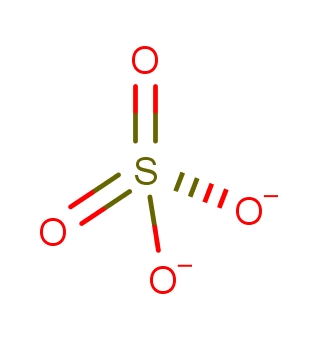
| CAS Number | 14808-79-8 |
|---|---|
| Molecular Formula | O4S |
| Molecular Weight | 96.060 |
| InChI Key | QAOWNCQODCNURD-UHFFFAOYSA-L |
| LogP | -2.20 |
| Synonyms |
|
Applications:
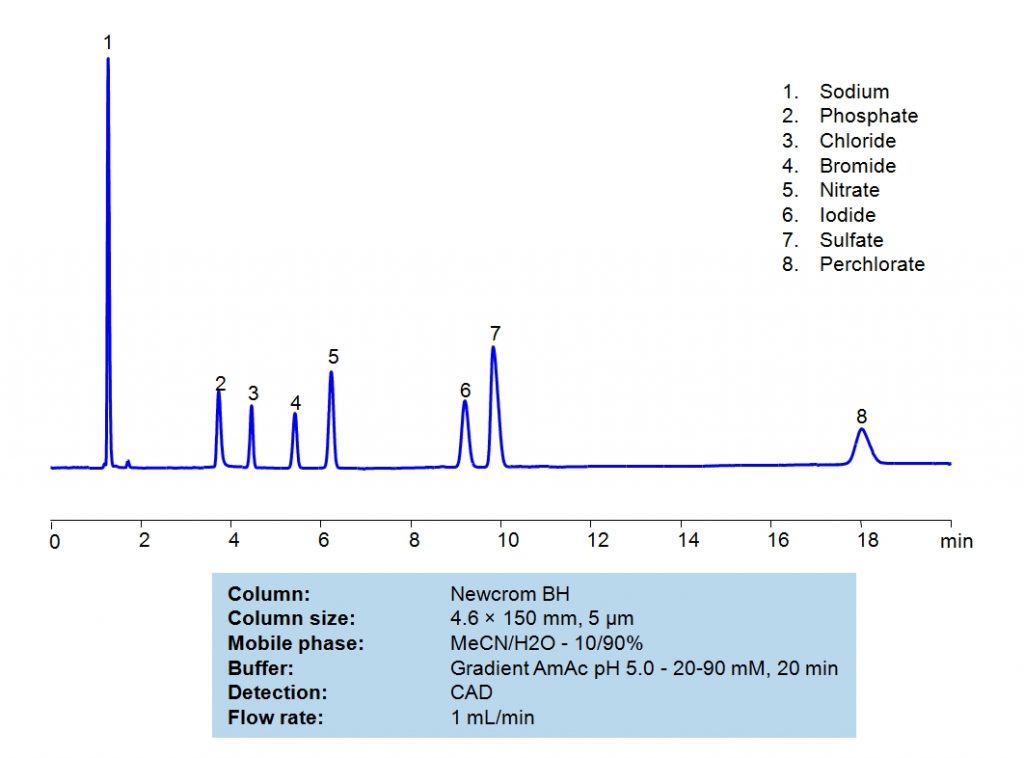
HPLC Separation of Inorganic Anions on Newcrom BH Column
October 23, 2019
| Column | Newcrom BH, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 10/90% |
| Buffer | Gradient AmAc pH 5.0 – 20-90 mM , 20 min |
| Flow Rate | 1.0 ml/min |
| Detection | CAD (Corona) (MS-compatible mobile phase) |
| Class of Compounds | Ions, Hydrophilic, Ionizable |
| Analyzing Compounds | Sodium, Phosphate, Chloride, Bromide, Nitrate, Iodide, Sulfate, Perchlorate |
Application Column
Newcrom BH
The Newcrom columns are a family of reverse-phase-based columns. Newcrom A, AH, B, and BH are all mixed-mode columns with either positive or negative ion-pairing groups attached to either short (25 Å) or long (100 Å) ligand chains. Newcrom R1 is a special reverse-phase column with low silanol activity.
Select optionsChloride
Iodide
Iodine
Nitrate
Perchlorate
Phosphate
Sodium
Sulfate

HPLC Analysis of Sulfate and Phosphate Ions on Primesep Columns
October 14, 2010
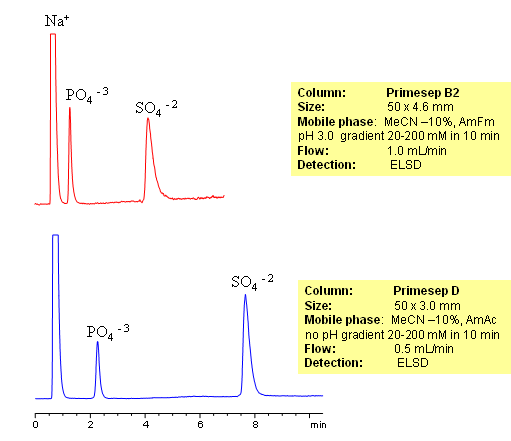
Inorganic and organic ions are usually analyzed by ion-exchange mechanism. In this application, we used short Primesep B2 and Primesep D mixed-mode HPLC columns to retain and separate phosphate and sulfate ions. Both ions are retained by ion-exchange mechanism. Detection technique for analysis of these anions is Evaporative Light-Scattering Detector (ELSD).
Application Column
Primesep B2
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsPrimesep C
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsPrimesep D
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsSulfate

HPLC Separation of Sulfamic and Sulfuric Acid on Primesep N HILIC Column
July 16, 2009
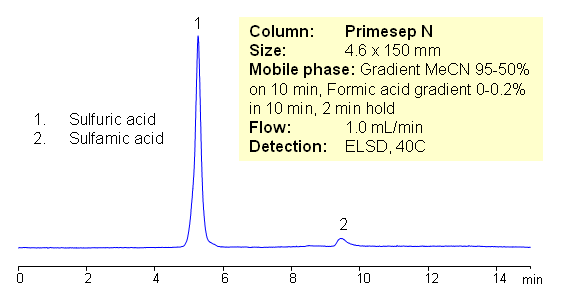
Sulfuric acid (sulfate ions) and sulfamic acid are analyzed in HILIC mode on Primesep N column. Both compounds are hydrophilic in nature and lack UV activity. Compounds are retained by hydrophilic interaction mechanism. Sulfate ion and sulfamic acid are monitored by ELSD. Method can be used for analysis of other inorganic ions.
Application Column
Primesep N
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsAminosulfonic Acid
Sulfamic Acid
Sulfate

Simultaneous Determination of Sodium and Sulfate Ions on Obelisc N Mixed-mode HILIC Column
July 16, 2009
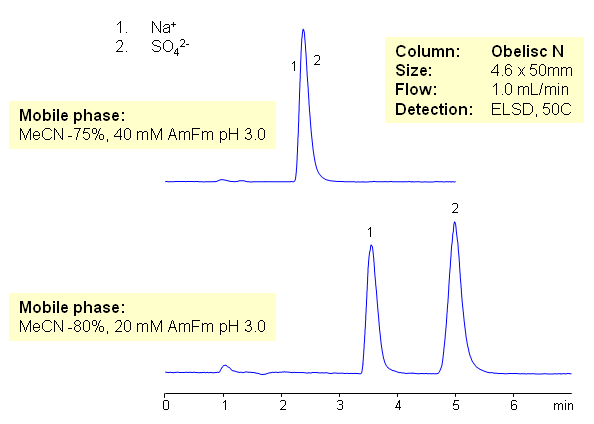
Inorganic cations and anions are usually analyzed by two different methods – cation-exchange and anion-exchange. This requires development and validation of two methods. Both ions can be analyzed on mixed-mode HILIC column with ELSD detection. Both ions are retained by combination of HILIC and ion-exchange mechanism.
| Column | Obelisc N, 4.6×50 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmFm, pH 3.0 |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Acid, Hydrophilic, Ionizable |
| Analyzing Compounds | Sodium, Sulfate Ions |
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsSulfate

HPLC Separation of Sulfate and Phosphate Ions on Mixed-Mode HPLC
December 6, 2007
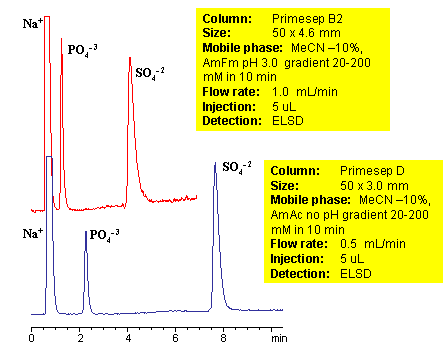
Sulfate and phosphate are separated on Primesep B2 and Primesep D column by anion-exchange mechanism. Because both sulfate and phosphate ions are not UV-active, ELSD is used to monitor separation of both anions. Method can be used to quantitate phosphate and sulfate in various pharmaceutical and chemical formulation, products and solution, drinking and ground water. Retention time is adjusted by increase or decrease of buffer concentrations. Two anions can be retained and separated on a very short column due to strong ion-exchange interaction. Method shows good reproducibility and versatility.
| Column | Primesep B2, 4.6×50 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmFm pH 3.0 |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Ions, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Sodium, Phosphate, Sulfate |
Application Column
Primesep B2
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsPrimesep D
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsSodium
Sulfate