| Synonyms |
|---|
Applications:
Retention of Taurine on Obelisc N Column
July 3, 2013
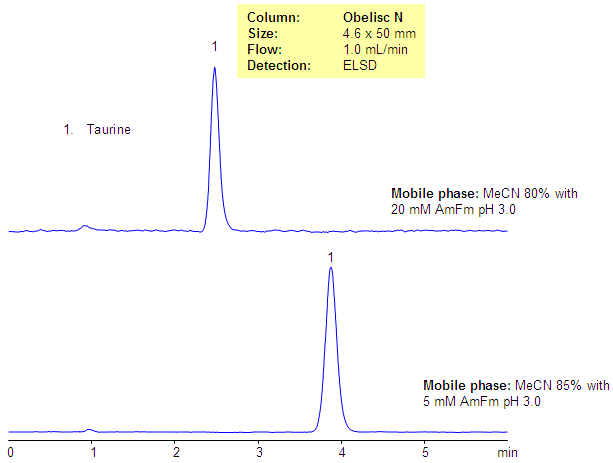
Taurine, or 2-aminoethanesulfonic acid, is a very polar zwitter-ionic compound. Taurine is used as an additive for various nutrition composition. The polar and zwitter-ionic nature of taurine prevent it from analysis by RP chromatography, in addition to that, taurine is not UV active and cannot be monitored by UV. The analytical method for analysis of taurine was developed on the Obelisc N HILIC/mixed-mode column. This method with some modifications can be used for the analysis of taurine in complex mixtures with a ELSD or a LC/MS detector.
| Column | Obelisc N, 4.6×50 mm, 5 µm, 100A |
| Mobile Phase | MeCN |
| Buffer | AmFm pH 3.0 |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Amino sulfonic acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Taurine |
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsZwitterion

Retention of Taurine on SHARC Column
July 3, 2013
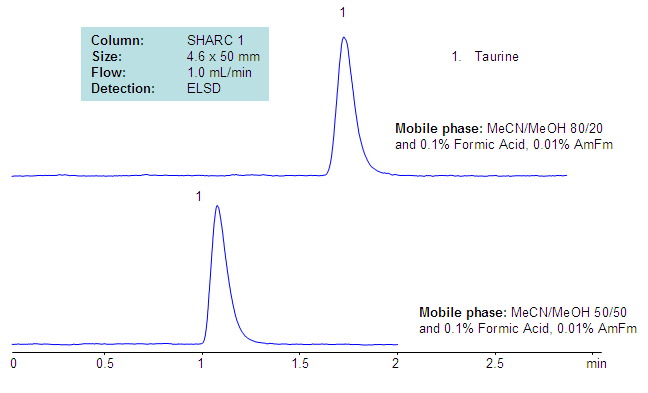
Taurine, or 2-aminoethanesulfonic acid, is a very polar zwitter-ionic compound. Taurine is used as an additive for various nutrition composition. The polar and zwitter-ionic nature of taurine prevent it from analysis by RP chromatography, in addition to that, taurine is not UV active and cannot be monitored by UV. The analytical method for analysis of taurine was developed on the SHARC 1 hydrogen-bonding column. This method with some modifications can be used for the analysis of taurine in complex mixtures with a ELSD or a LC/MS detector.
| Column | Sharc 1, 4.6×50 mm, 5 µm, 100A |
| Mobile Phase | MeCN/MeOH |
| Buffer | Formic Acid – 0.1%, AmFm – 0.01% |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Amino sulfonic acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Taurine |
Application Column
SHARC 1
The SHARC™ family of innovative columns represents the first commercially available columns primarily utilizing separation based on hydrogen bonding. SHARC stands for Specific Hydrogen-bond Adsorption Resolution Column. Hydrogen bonding involves an interaction or attraction between a bound hydrogen atom and molecules containing electronegative atoms, such as oxygen, nitrogen, and fluorine.
Select optionsZwitterion

HPLC Separation of Glutamic Acid and GABA
July 8, 2011
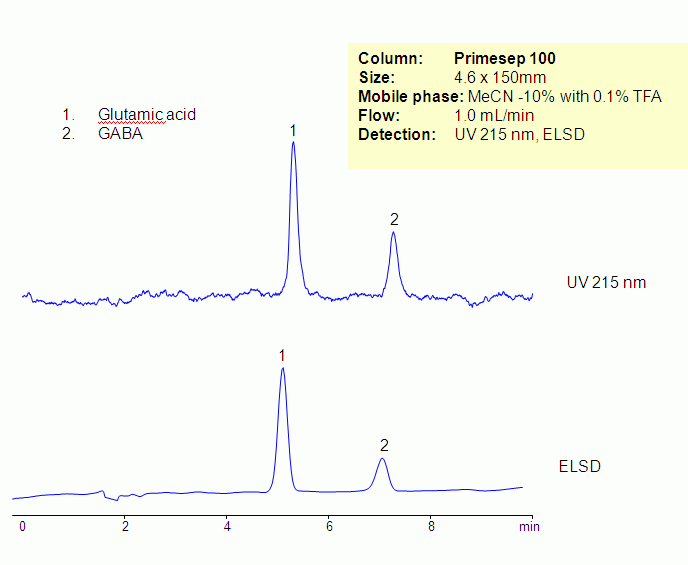
Glutamic acid and GABA are neurotransmitters. Glutamic acid and GABA are non-essential amino acids. They are hydrophilic and zwitter-ionic in nature . At lower pH, carboxylic acid groups of amino acids are not ionized, making them more hydrophobic and basic. Underivatized glutamic acid and GABA were retained and separated on a Primesep 100 column using ACN/water/TFA mobile phase. Amino acids can be monitored by low UV or ELSD/CAD. Retention is provided by reversed-phase and cation-exchange mechanism. Method can be used for analysis of underivatized amino acids in various matrices including supplements, vitamin and other complex mixtures. various mobile phase can be used with corresponding detection techniques.
| Column | Primesep 100, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 10/90% |
| Buffer | TFA – 0.1% |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 215 nm, ELSD |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Glutamic acid, GABA |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsZwitterion
gamma-Aminobutyric Acid (GABA)
UV Detection

HPLC Separation of Morpholino Sulfates
November 21, 2010
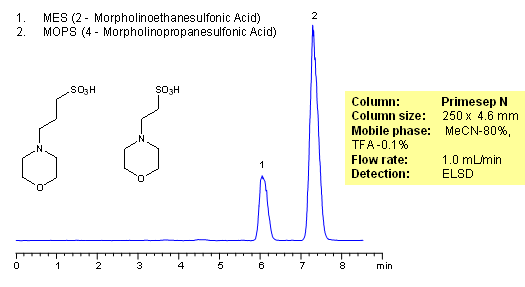
MOPS and MES are buffers used in biology and biochemistry. They consist of a morpholine ring and propanesulfonic acid. Both molecules are zwitter-ionic and very polar. No retention can be achieved on reversed-phase columns. MOPS and MES are analyzed in HILIC mode on a Primesep N HPLC column. Other biological buffers can be analyzed with this general method. Biological buffers are not UV active and can be monitored by ELSD, CAD or LC/MS.
Application Column
Primesep N
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options4-Morpholinepropanesulfonic Acid (MOPS)
Zwitterion

HPLC Separation of Acidic, Basic and Zwitterionic Compounds on Special Columns for Retention of Polar Compounds
April 10, 2010
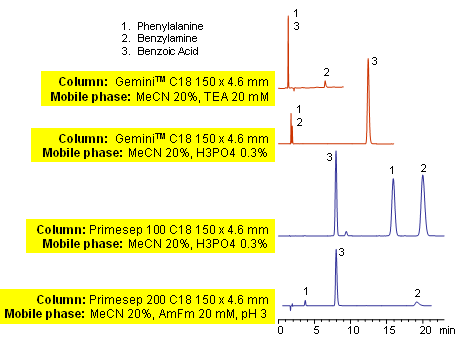
Primesep mixed-mode columns are designed to separate basic, acidic, neutral and zwitter-ionic compounds. Elution of these compounds is facilitated by acetonitrile and ions in the mobile phase. In all cases the retention time on mixed-mode columns is significantly longer than on traditional reverse-phase columns. Presence of cation-exchange mechanism in Primesep 100 column allows to achieve better retention and peak shape for analytes like phenylalanine and benzylamine. Various buffers can be used along with different detection techniques. This HPLC method can be adopted as a universal approach for analysis of basic, acidic and zwitter-ionic compounds in complex mixtures and formulations. Compounds can be monitored by UV, ELSD, CAD and LC/MS.
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsBenzylamine
Phenylalanine
Zwitterion

Comparison of Obelisc N, SeQuant?® HILIC, and Bare Silica
March 3, 2010
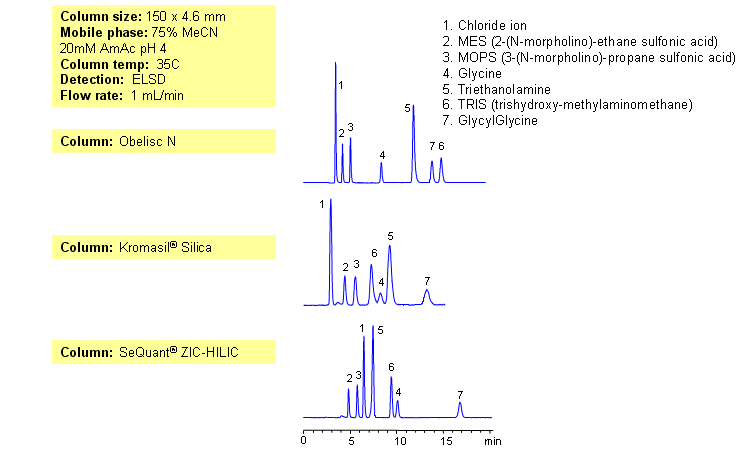
Obelisc N mixed-mode columns provide different selectivity than other HILIC columns — presence of ion-exchange mechanism contributes to a different selectivity. Depending on the pH of the mobile phase, ion-exchange mechanism can be enhanced or suppressed. Two amino acids (glycine and glycylglycine), two basic compounds (triethanolamine and TRIS) and two zwitter-ionic compounds (MOPS and MES) are separated by combination of HILIC and ion-exchange mechanisms. Compounds elution can be monitored by Evaporative Light-Scattering Detector (ELSD), Corona (CAD), LC/MS or other detection techniques.
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select options
HPLC Separation of alpha-Aminobutyric, beta-Aminobutyric, and gamma-Aminobutyric acids on Obelisc N
March 3, 2010
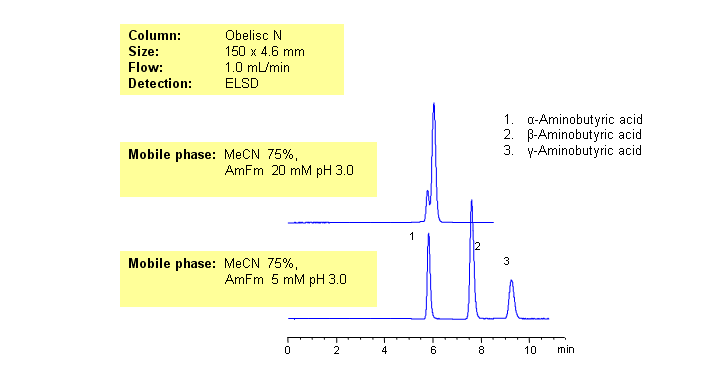
GABA (neurotransmitter) and its isomers are polar zwitter-ionic compounds. Due to the position of amino-groups, all three compounds show different polar and basic properties. The isomers of aminobuturic acid are separated on an Obelisc N HILIC/cation-exchange column. Buffer concentration has a different effect on retention of alpha-, beta-, and gamma-aminobutyric acid. This general and robust method can be used for separation of other polar and ionizable compounds and isomers by mixed-mode chromatography.
| Column | Obelisc N, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmFm pH 3.0 |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD |
| Class of Compounds |
Acid |
| Analyzing Compounds | Alpha-Aminobutyric acid, Beta-Aminobutryic acid, Gamma-Aminobutyric acid (GABA) |
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsalpha-Aminobutyric Acid
beta-Aminobutyric Acid
gamma-Aminobutyric Acid (GABA)

Effect of Buffer on HPLC Separation of Buffers
August 22, 2008
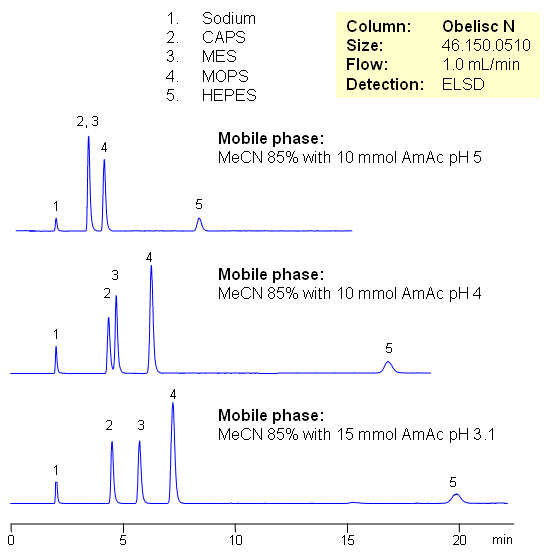
HEPES, CAPS, MES and MOPS are zwitterionic organic chemical buffering agents. These are very polar compounds which are not retained by traditional reverse phase chromatography. These compounds are zwitterions in nature and can be separated by mixed-mode hydrophilic interaction chromatography on Obelisc N column. Retention is achieved by combination of HILIC and ion-exchange mechanisms. These buffering agents do not have UV active groups, but can be analyzed with ESLD, LC/MS, and CAD detection.
Application Column
Obelisc N
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select options4-Morpholinepropanesulfonic Acid (MOPS)
CAPS (3-(Cyclohexylamino)propanesulfonic acid)
HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic Acid)
Zwitterion

HPLC Separation of Taurine
December 6, 2007
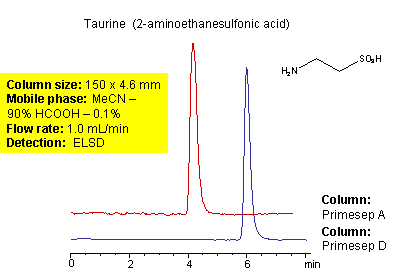
Taurine (2-aminoethanesulfonic acid) is organic zwitterionic compound. Taurine is a non-essential sulfur-containing amino acid that functions with glycine and gamma-aminobutyric acid as a neurotransmitter. Taurine is incorporated into one of the most abundant bile acids, chenodeoxycholic acid, where it serves to emulsify dietary lipids in the intestine, promoting digestion. It is used in food and pharmaceutical formulations. High polarity and zwitterionic nature of taurine complicates analysis of this compound by reverse-phase chromatography. Two methods for the analysis of taurine are developed on mixed-mode columns. Taurine is retained on Primesep A column by HILIC cation-exchange mechanism and on Primesep D column by HILIC anion-exchange mechanism. Method can be used for fast and effective quantitation of taurine in various products including energy drinks. Method requires ELSD or LC/MS detection due to lack of UV activity for taurine.
Application Column
Primesep D
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsPrimesep A
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsZwitterion

Retention Characteristics of Betaine in Mixed-Mode HPLC
May 5, 2005
The retention of the zwitterion betaine on Primesep 200 demonstrates the tunability of Primesep columns. Retention of betaine is altered by acid type, amount of acid in the mobile phase, and the amount of organic solvent. Typical reversed-phase columns do not show this tunability with simple mass spec compatible mobile phases of water, acetonitrile (MeCN, ACN) and trifluoracetic acid (TFA) with evaporative light scattering detection (ELSD).
Application Column
Primesep 200
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsQuaternary Amines
Zwitterion

TFA Concentration Effect on Retention of Tricine
April 5, 2004
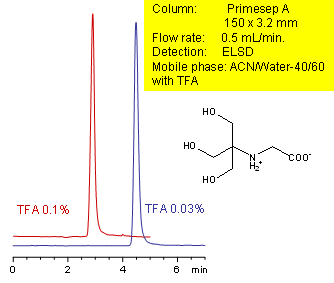
Primesep A retains the hydrophilic zwitterion tricine by a combination of polar interactions and ion exchange. Tricine is not retained by traditional reversed-phase chromatography. The retention on Primesep A is adjustable by changing the concentration of TFA in the mobile phase. Excellent peak shape results with a mass spec compatible mobile phase of water, acetonitrile (MeCN, ACN) and trifluoracetic acid (TFA) with evaporative light scattering detection (ELSD).
Application Column
Primesep A
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsZwitterion









