| CAS Number | 61336-70-7 |
|---|---|
| Molecular Formula | C16H25N3O8S |
| Molecular Weight | 419.450 |
| InChI Key | MQXQVCLAUDMCEF-CWLIKTDRSA-N |
| LogP | 0.601 |
| Synonyms |
|
Applications:
HPLC Separation of β-Lactam Antibiotics on Primesep 100 Column
March 18, 2021
Separation type: Liquid Chromatography Mixed-mode
View on hplc.cloud
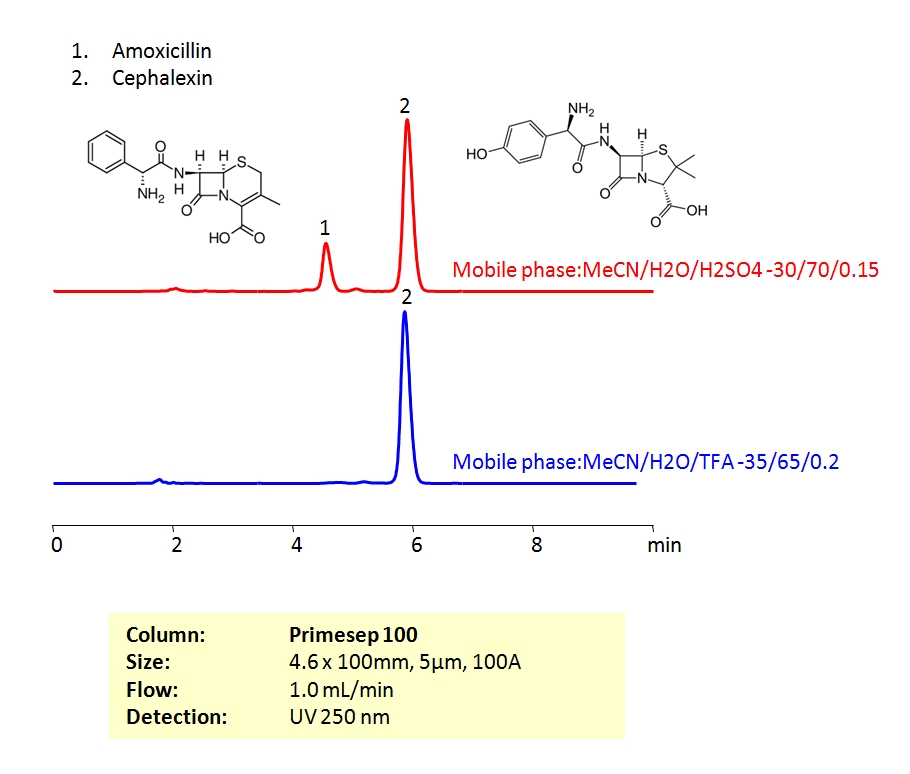
High Performance Liquid Chromatography (HPLC) Method for Analysis of Amoxicillin and Cephalexin.
Both amoxicillin and cephalexin are beta-lactam antibiotics. Beta-lactam antibiotics contain a beta-lactam ring in their molecular structure. As a group, these drugs are active against many gram-positive, gram-negative and anaerobic organisms. Both compounds can be retained and baseline separated with an isocratic method in 30/70 Acetonitrile (ACN) and water mobile phase with a Sulfuric acid (H2SO4) buffer. UV detection at 250 nm.
| Column | Primesep 100, 4.6×100 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | TFA, H2SO4 |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 250 nm |
| Class of Compounds |
Drug, Antibiotics, Hydrophobic, Ionizable |
| Analyzing Compounds | Cephalexin, Amoxicillin |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsAmoxicillin hydrate (1:3)
Cephalexin

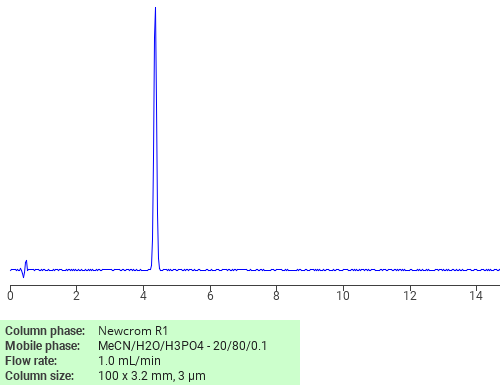
Separation of Amoxicillin hydrate (1:3) on Newcrom R1 HPLC column
February 16, 2018
Amoxicillin hydrate (1:3) can be analyzed by this reverse phase (RP) HPLC method with simple conditions. The mobile phase contains an acetonitrile (MeCN), water, and phosphoric acid. For Mass-Spec (MS) compatible applications the phosphoric acid needs to be replaced with formic acid. Smaller 3 µm particles columns available for fast UPLC applications. This liquid chromatography method is scalable and can be used for isolation impurities in preparative separation. It also suitable for pharmacokinetics.
Application Column
Newcrom R1
The Newcrom columns are a family of reverse-phase-based columns. Newcrom A, AH, B, and BH are all mixed-mode columns with either positive or negative ion-pairing groups attached to either short (25 Å) or long (100 Å) ligand chains. Newcrom R1 is a special reverse-phase column with low silanol activity.
Select options