| CAS Number | 113-00-8 |
|---|---|
| Molecular Formula | CH5N3 |
| Molecular Weight | 59.072 |
| InChI Key | ZRALSGWEFCBTJO-UHFFFAOYSA-N |
| LogP | -1.45 |
| Synonyms |
|
Applications:
HPLC Application for Retention of Guanidine by Mixed-Mode Chromatography
December 6, 2007

Guanidine is highly basic (pKa 12.5), and a polar compound is formed by the oxidation of guanine. In the human body, guanine is a byproduct of protein metabolism and can be found in urine. Guanine can be retained by the cation-exchange mechanism on Primesep 100 column. Guanine is slightly UV active and can be observed at 200 nm. A better detection technique is ELSD, which allows you to see low concentrations of guanine. The temperature of the ELSD must be maintained relatively low to insure proper sensitivity of the method. This method can be used for analysis of other guanidine-based compounds. If substituted guanidine has hydrophobic properties, it will retain based on ion-exchange and reverse phase mechanisms.
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsUV Detection

HPLC Separation of Guanidine and Benzoic Acids on Primesep 100 Column
October 4, 2007
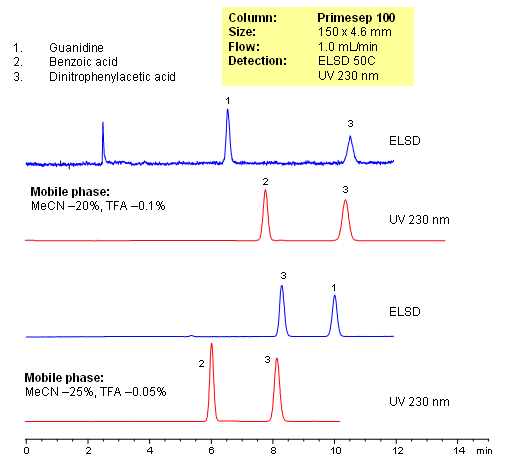
| Column | Primesep 100, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | TFA |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 230 nm, ELSD |
| Class of Compounds |
Acid, Hydrophilic, Ionizable |
| Analyzing Compounds | Guanidine, Benzoic acid, Dinitrophenylacetic Acid |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsDinitrophenylacetic Acid
Guanidine
UV Detection

Separation of Guanidine on Primesep 100 Column
October 4, 2005
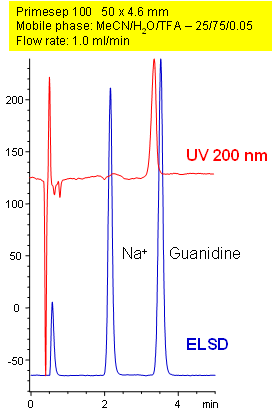
Primesep 100 separates guanidine from its sodium counterion on a short 50 mm column by cation exchange. Guanidine is found in urine as a product of protein metabolism and is also used in the manufacture of plastics and explosives. Guanidine is detected by UV detection at 200 nm, but UV cannot detect the sodium ion. If an evaporative light scattering detector (ELSD) is used, both components can be detected. The separation method uses a mobile phase mixture of water, acetonitrile (MeCN, ACN) and trifluoroacetic acid (TFA).
| Column | Primesep 100, 4.6×50 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | TFA |
| Flow Rate | 1.0 ml/min |
| Detection | ELSD, UV 200nm |
| Class of Compounds |
Ions, Hydrophilic, Ionizable |
| Analyzing Compounds | Sodium, Guanidine |
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsSodium
UV Detection