| CAS Number | 59-42-7 |
|---|---|
| Molecular Formula | C9H13NO2 |
| Molecular Weight | 167.208 |
| InChI Key | SONNWYBIRXJNDC-VIFPVBQESA-N |
| LogP | -0.310 |
| Synonyms |
|
Applications:
HPLC Method for Analysis of Neurotransmitters Phenylephrine, Epinephrine and Norepinephrine on BIST™B+ Column
July 14, 2022
| Separation type: Bridge Ion Separation Technology, or BIST™ | ||||||||||||||
| BIST™ Ionic Modifier Preparation | ||||||||||||||
| High Performance Liquid Chromatography (HPLC) Method for Analysis of Phenylephrine, Epinephrine, Norepinephrine
Epinephrine and norepinephrine are key neurotransmitters and adrenal hormones naturally produced by the body to generate the fight-or-flight response, and synthesized by pharmaceutical companies to treat a host of issues, such as allergic reactions and low blood pressure. Phenylephrine is a related, synthetic compound made to treat low blood pressure, congestion, and hemorrhoids. Using SIELC’s newly introduced BIST™ method, these three similar neurotransmitters, which protonate in water, can be retained on a positively-charged anion-exchange BIST™ B+ column. There are two keys to this retention method: 1) a multi-charged, negative buffer, such as Sulfuric acid (H2SO4), which acts as a bridge, linking the positively-charged amine analytes to the positively-charged column surface and 2) a mobile phase consisting mostly of organic solvent to minimize the formation of a solvation layer around the charged analytes. Using this new and unique analysis method, Epinephrine, norepinephrine, and phenylephrine can be retained and UV detected at 200 nm.
|
||||||||||||||
|
Application Column
BIST B+
BIST™ columns offer a unique and effective way to achieve separations that were traditionally challenging or even impossible with other HPLC columns. With the use of a special mobile phase, these ion exchange columns provide very strong retention for analytes with the same charge polarity as the stationary phase, unlocking new chromatography applications. What makes BIST™ columns stand out is their proprietary surface chemistry, which results in superior selectivity, resolution, and sensitivity. These columns offer a simple, efficient solution for a variety of analytical challenges, making them an excellent choice for researchers and analysts across many different fields. To learn more about the technology that powers BIST™ columns and to explore related applications, check out https://BIST.LC.
Select optionsNorphenylephrine
Phenylephrine
Phenylephrine hydrochloride

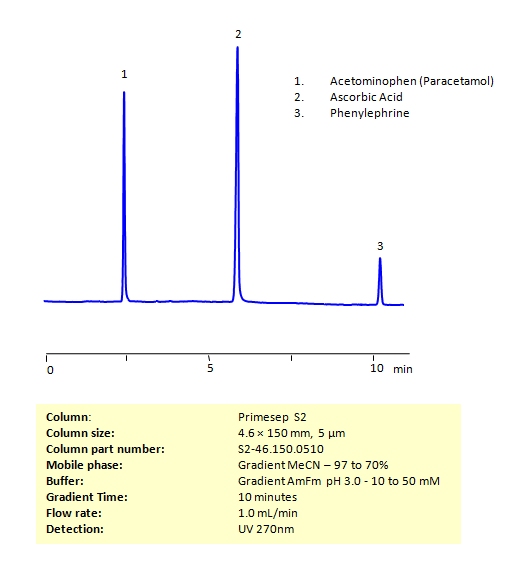
HPLC Method for the Determination of Acetaminophen, Phenylephrine, and Ascorbic Acid on Primesep S2 Column
October 5, 2021
Separation type: Liquid Chromatography Mixed-mode
High Performance Liquid Chromatography (HPLC) Method for Analysis of Acetaminophen, Phenylephrine, and Ascorbic Acid
Acetaminophen (or paracetamol) is one of the most popular over-the-counter painkillers all over the world (in the US it is best known under the brand name Tylenol). Ascorbic acid (also known as Vitamin C) helps your body build up muscle, cartilage, and other important building blocks in your body. Phenylephrine is a common over-the-counter decongestant, but can also be used for pupil dilation and hemorrhoid treatment. While all of these compounds are available at your local pharmacy, Vitamin C intake can reduce the body’s ability to break down acetaminophen.
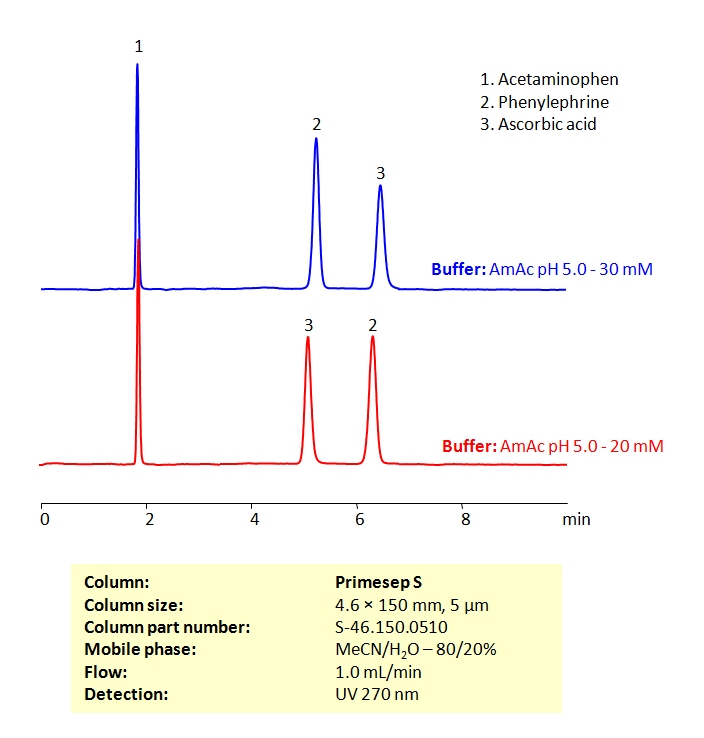
All three of these compounds can be measured at low UV. Using a Primsep S normal phase column and a mobile phase consisting of water and acetonitrile (MeCN) with an Ammonium acetate (AmAc) buffer, these three compounds can be separated and UV detected at 270 nm. Varying the buffer concentration changes the order in which phenylephrine and ascorbic acid are retained. This method is compatible with Mass Spectrometry.
| Column | Primesep S2, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | Gradient MeCN – 97 to 70% |
| Buffer | Gradient Ammonium Formate pH 3.0 – 10 to 50 mM |
| Flow Rate | 1.0 ml/min |
| Detection | UV 270 nm MS- compatible mobile phase |
| Class of Compounds | Drug, Acid |
| Analyzing Compounds | Acetaminophen, Phenylephrine, and Ascorbic Acid |
Application Column
Primesep S2
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsAscorbic Acid
Phenylephrine
Phenylephrine hydrochloride

HPLC Method for the Determination of Acetaminophen, Phenylephrine, and Ascorbic Acid on Primesep S Column
October 5, 2021
Separation type: Liquid Chromatography Mixed-mode
High Performance Liquid Chromatography (HPLC) Method for Analysis of Acetaminophen, Phenylephrine, and Ascorbic Acid
| Column | Primesep S, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O – 80/20% |
| Buffer | Ammonium acetate pH 5.0 |
| Flow Rate | 1.0 ml/min |
| Detection | UV 270nm MS- compatible mobile phase |
| Class of Compounds | Drug |
| Analyzing Compounds | Acetaminophen, Phenylephrine, and Ascorbic Acid |
Application Column
Primesep S
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsAscorbic Acid
Phenylephrine
Phenylephrine hydrochloride

HPLC Separation of Pseudoephedrine, Norephedrine, Phenylephrine, and Norphenylephrine Using Hydrogen Bonding Mode
June 15, 2012
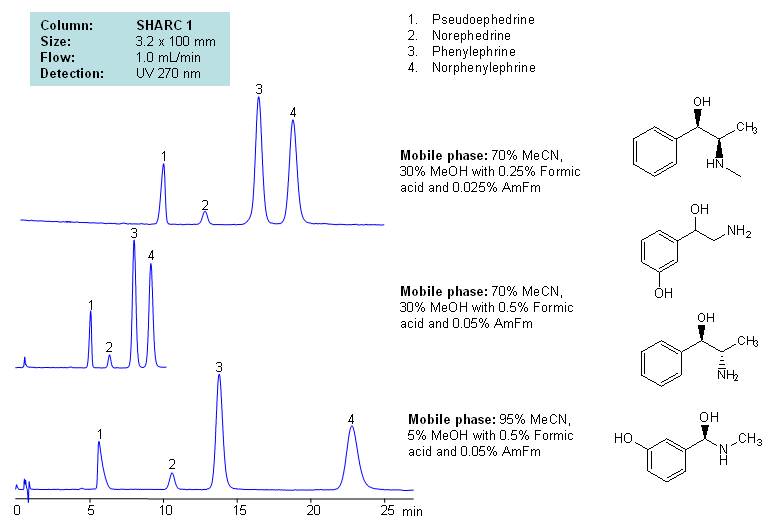
Application Notes: Neurotransmitters are polar basic compounds. Several HPLC techniques are used to analyze these hydrophilic molecules. The most common techniques are reversed-phase with ion-pairing reagents chromatography, ion chromatography, HILIC, and mixed-mode chromatography. SIELC developed a new approach of analysis, which is based on hydrogen bond interactions. Hydrogen-bonding offers unique and alternative selectivity to traditional approaches like reversed-phase, HILIC, ion-exchange, and mixed-mode chromatography. Our methods are fully compatible with ELSD, LC/MS and prep chromatography.
Application Columns: SHARC 1, 3.2×100 mm, 5 um, 100A. To learn more about SHARC 1 columns click here. To order this column click here. To see more chromatographic separations check our web site.
Application compounds: Pseudoephedrine, norephedrine, phenylephrine and norphenylephrine
Detection technique: UV, LC/MS
| Column | Sharc 1, 3.2×100 mm, 5 µm, 100A |
| Mobile Phase | MeCN/MeOH |
| Buffer | AmFm, Formic acid |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Pseudoephedrine, Norephedrine, Phenylephrine, Norphenylephrine |
Application Column
SHARC 1
The SHARC™ family of innovative columns represents the first commercially available columns primarily utilizing separation based on hydrogen bonding. SHARC stands for Specific Hydrogen-bond Adsorption Resolution Column. Hydrogen bonding involves an interaction or attraction between a bound hydrogen atom and molecules containing electronegative atoms, such as oxygen, nitrogen, and fluorine.
Select optionsNorphenylephrine
Phenylephrine
Pseudoephedrine (PSE)

HPLC Separation of Phenylephrine with Tannic Acid Formulation
November 21, 2010
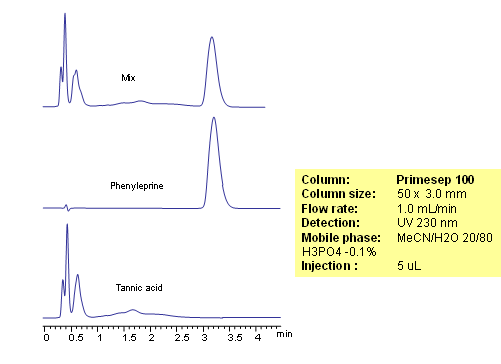
Phenylephrine is a hydrophilic basic compound. Phenylephrine is used primarily as a decongestant in cough composition. Tannic acid is poly-phenolic compound. Tannic acid is also used to produce tannate salts of certain antihistamins and anti-tussives to impart increased stability or slow release properties to the API (active pharmaceutical ingredient). Phenylephrine in the presence of tannic acid is analyzed by mixed-mode HPLC on a Primesep 100 column. Retention time for phenylephrine is controlled by the amount of acetonitrile and buffer concentration. Tannic acid elutes closer to void and not interferes with analysis of phenylephrine. Method can be used with UV detection.
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsTannic Acid

HPLC Separation of Caffeine and Phenylephrine
October 14, 2010
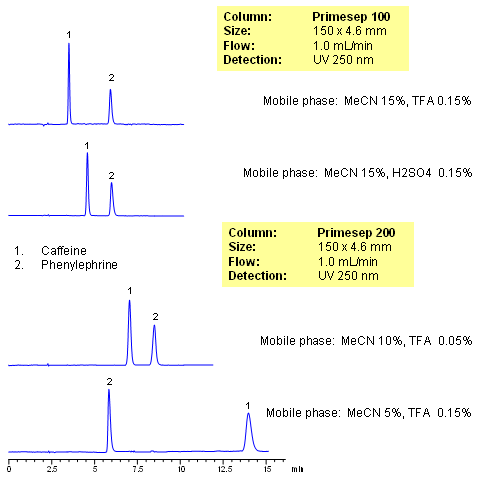
Caffeine and phenylephrine are some of the components of various pain killer/fever reducers/cough compositions. In this application, both of these compounds are separated on Primesep 100 and Primesep 200 columns. Caffeine is retained by reversed-phase mechanisms and phenylephrine is retained by combination of reversed-phase and cation-exchange mechanisms. Retention time for caffeine is controlled by the amount of acetonitrile, while retention time of phenylephrine is controlled by amount of the acetonitrile, buffer concentration and buffer pH. The method can be used for analysis of components of pain, cough and cold medication in pharmaceutical production. Method is robust and reproducible and provides good retention and peak shape. Compounds are monitored by UV, ELSD, CAD or LC/MS. Analysis of active components on biofluids (urine, plasma, blood, etc) is possible with additional sample preparation (protein precipitation, SPE, etc.)
Application Column
Primesep 100
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsPrimesep 200
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsPhenylephrine

HPLC Separation of Polar Drugs with MS-compatible Method
October 12, 2010
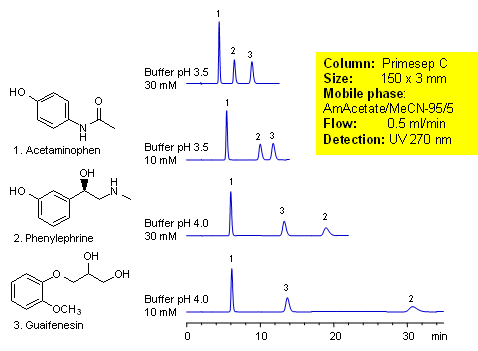
Acetaminophen is an over-the-counter pain and fever reducer, and a major component of cold and flue remedies. Phenylephrine is a decongestant. Guaifenesin is a mucolytic agent used to relieve respiratory difficulties. These three compounds of cough medication were separated by mixed-mode chromatography on a Primesep C HPLC column. Retention and order of elution for phenylephrine can be changed by buffer concentration and buffer pH. Method can be used for analysis of cough and cold composition during the production and in QC/QA environment. Method is compatible with LC/MS and can be used to analyze these components in biological fluids. This generic HPLC method is robust and reproducible.
| Column | Primesep C, 3×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN |
| Buffer | AmAc |
| Flow Rate | 0.5 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds |
Drug, Analgetic, Acid, Hydrophilic, Ionizable, |
| Analyzing Compounds | Acetaminophen, Pseudoephedrine, Guaifenesin |
Application Column
Primesep C
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsGuaifenesin
Phenylephrine

Effect of pH on Retention of Basic Compounds on Primesep C Columns
January 13, 2010
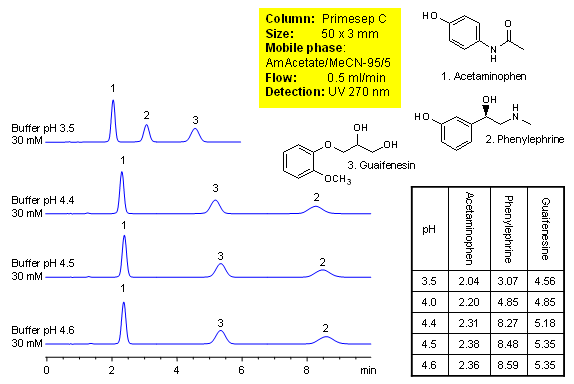
Method for separation of components of cough medication shows how retention for acetaminophen, phenylephrine and guaifenesin is controlled. Order of elution for compounds can be changed based on composition of the mobile phase. The method is compatible with UV, ELSD and LC/MS and can be used for quantitation of drugs in formulation. Primesep C mixed-mode HPLC column is ideal candidate for analysis of cough compositions. Basic compounds are well retained in mixed-mode chromatography without use of ion-pairing reagents. Several companies validated this approach. The method is reproducible and robust and can be used in both production environments and R&D. This HPLC method can be adopted as general approach for analysis of basic compounds in various mixtures.
| Column | Primesep C, 3×50 mm, 5 µm, 100A |
| Mobile Phase | MeCN |
| Buffer | AmAc |
| Flow Rate | 0.5 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds |
Drug, Analgetic, Acid, Hydrophilic, Ionizable, |
| Analyzing Compounds | Acetaminophen, Pseudoephedrine, Guaifenesin |
Application Column
Primesep C
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsGuaifenesin
Phenylephrine

HPLC Separation of Neurotransmitters and Related Drugs
August 22, 2008
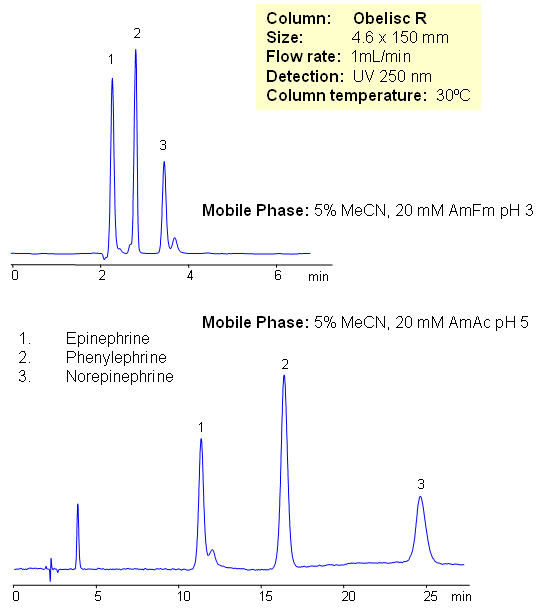
Epinephrine and norepinephrine (adrenaline and noradrenaline) are hormones and neurotransmitters. Epinephrine is synthesized from norepinephrine in a synthetic pathway shared by all catecholamines, including L-dopa, dopamine, norepinephrine, and epinephrine. Phenylephrine is used as a decongestant, available as an oral medicine or as a nasal spray. Phenylephrine is now the most common over-the-counter (OTC) decongestant. All three compounds are used in various drug compositions. Separation of epinephrine and norepinephrine is a challenging task due to polarity and close properties of two compounds. Epinephrine, norepinephrine and phenylephrine are separated in this method on Obelisc R mixed-mode HPLC columns. The method is very sensitive to variation of pH and pH adjustment can be used to achieve desired selectivity and retention time. Other catecholamines can be analyzed using this HPLC method. The method can be used as a stability indicating or a impurity profiling approach to the analysis of neurotransmitters in drug formulation, blood, serum and urine.
| Column | Obelisc R, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | AmFm |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 250 nm |
| Class of Compounds |
Drug, Acid, Hydrophilic, Ionizable, Hormone |
| Analyzing Compounds | Phenylalanine, Norepinephrine, Epinephrine |
Application Column
Obelisc R
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Select optionsNorepinephrine
Phenylephrine

HPLC Separation of Acetaminophen, Phenylephrine, and Diphenhydramine
March 3, 2006
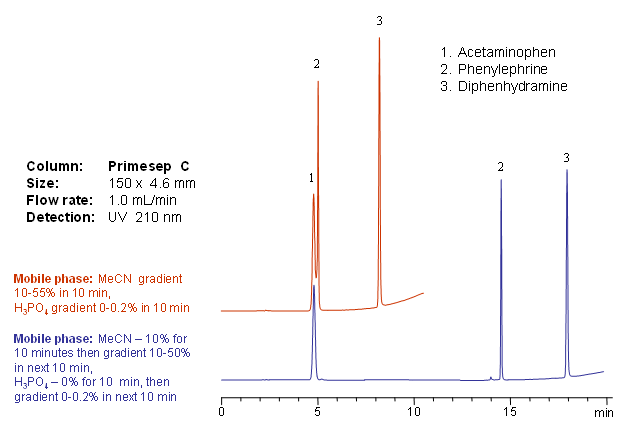
Primesep C separates acetaminophen (paracetamol), phenylephrine, and diphenhydramine– common over-the-counter cold and flu medication ingredients such as Tylenol products—by either an acetonitrile or phosphoric acid gradient. The HPLC separation uses a mobile phase of water, acetonitrile (MeCN, ACN), and phosphoric acid with ultraviolet (UV) detection at 210 nm.
| Column | Primesep C, 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN |
| Buffer | H3PO4 |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 210 nm |
| Class of Compounds |
Drug, Analgetic, Acid, Hydrophilic, Ionizable, |
| Analyzing Compounds | Acetaminophen, Pseudoephedrine, Diphenhydramine |
Application Column
Primesep C
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select optionsDiphenhydramine
Phenylephrine