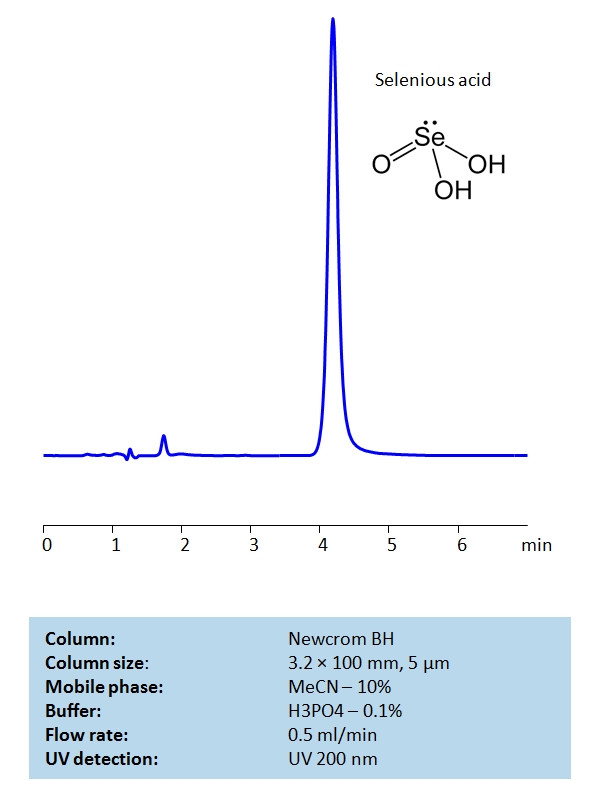
Selenious Acid is used in protecting and changing the color of steel, most often in weaponry. It is highly toxic in excessive quantities, and if ingested, it could lead to death. The Acid is also capable of penetrating the skin and can produce acute poisonings. Newcrom BH, a reverse-phase column, contains embedded basic ionizable groups and can retain Selenious Acid. The method is UV compatible and can be used as a general approach for analyzing similar compounds.
| Column | Newcrom BH, 3.2 x 100 mm, 5 µm, 100 A, dual ended |
| Mobile Phase | MeCN/H2O – 10/90% |
| Buffer | H3PO4 – 0.1% |
| Flow Rate | 0.5 ml/min |
| Detection | UV, 200 nm |
| Class of Compounds |
Acid, Hydrophilic, Ionizable |
| Analyzing Compounds | Selenious Acid |
Application Column
Newcrom BH
Column Diameter: 3.2 mm
Column Length: 100 mm
Particle Size: 5 µm
Pore Size: 100 A
Column options: dual ended