New more reliable method for EDTA analysis is available
EDTA (Ethylenediaminetetraacetic acid) is amino acid based compound which is widely used in industrial cleaners, detergents, photography, agrochemicals, textile, and food industries. EDTA serves as preservative in packaged food, vitamins and pharmaceutical formulations. It is used in organic and analytical laboratories as scavenger of metals, buffer solution, complexometric titration, masking agent for metal determination, etc.
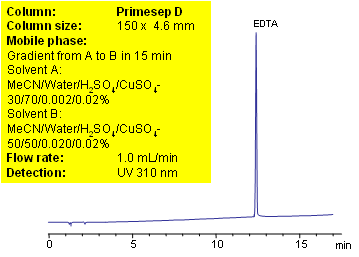
EDTA is very polar compound with strong chelating properties. Low UV activity and strong binding to metal ions makes HPLC analysis of EDTA very difficult. Reproducible method for determination of wide range on concentration of EDTA in various formulations is developed using Primesep D anion-exchange mixed mode column. Copper sulfate is used for visualization purposes to increase sensitivity of the method. EDTA forms UV active complex with some metal ions. This method can be used for highly accurate quantitation of EDTA in different mixtures and composition. Method demonstrates controllable retention and perfect peak shape. Equilibration of the column prior to analysis requires special attention.
If multiple injections produce different retention times, your column might not be equilibrated properly. Primesep D columns have a large ionic capacity towards anions. The shipping solvent for the Primesep D columns is ACN/water/TFA. The end point of gradient has only 4 mmol of sulfate ions. It takes over 5 hours to equilibrate a column with such low concentration. Before running your experiments you need to wash your column with 20% ACN and 0.2% of sulfuric acid for 1 hour. After you replace previous buffer/additive, you can set up your experiment. If you don’t change ionic modifier in your mobile phase the equilibration time is equivalent of 4-5 column volumes.
| Column | Primesep D , 4.6×150 mm, 5 µm, 100A |
| Mobile Phase | MeCN/H2O |
| Buffer | H2SO4 with CuSO4 |
| Flow Rate | 1.0 ml/min |
| Detection | UV 310 nm |
| Class of Compounds |
Acid |
| Analyzing Compounds | EDTA, or Ethylenediaminetetraacetic acid |
Application Column
Primesep D
The Primesep family of mixed-mode columns offers a wide variety of stationary phases, boasting unprecedented selectivity in the separation of a broad array of chemical compounds across multiple applications. Corresponding Primesep guard columns, available with all stationary phases, do not require holders. SIELC provides a method development service available to all customers. Inquire about our specially-tailored custom LC-phases for specific separations.
Select options




